About CEL-SCI
CEL-SCI (NYSE:CVM) is a small-cap market (~$500 million) clinical-stage cancer biopharmaceutical company. It has two products in the pipeline: Multikine and LEAPS. We will only be discussing Multikine in this article.
Multikine, a cocktail combination of cytokines and chemokines, is a prospective neoadjuvant treatment and an investigational drug candidate in Phase III event-driven clinical development for newly diagnosed advanced primary head and neck cancer.
The goal of Multikine is to boost the body's immune system prior to Standard of Care ("SOC"). Multikine is given right after diagnosis, before surgery, radiation and chemotherapy have destroyed the immune system. The event-driven Phase III study was fully enrolled with 928 patients and the last patient was treated in September 2016.
To prove an Overall Survival ("OS") benefit, the study required CEL-SCI to wait until 298 event (deaths) to have occurred among the two main comparator groups.
The nearly 10 year journey ended in April. On May 4, CEL-SCI announced that the 298th event occurred, ending the Phase III trial of Multikine.
The database is now being prepared for database lock. Once the database has been locked the final analysis of the trial results can be performed. CEL-SCI will continue to remain blinded to the study results throughout this process. CEL-SCI will be advised of the results when the analysis is completed and the study results will be announced to the public and investors at that time.
The CROs involved in study management are generating the remaining queries, performing source data verification, completing medical review, and building the final database needed to produce the final tables, listings, and figures."
It was a journey filled with many ups and downs.
Data is Now Locked
After nearly 7 months and a full medical review, data is now locked.
On December 7 CEL-SCI announced that "Data lock has already been completed."
Despite Geert saying numerous times that he wouldn't announce Data Lock, guess what, he announced it!
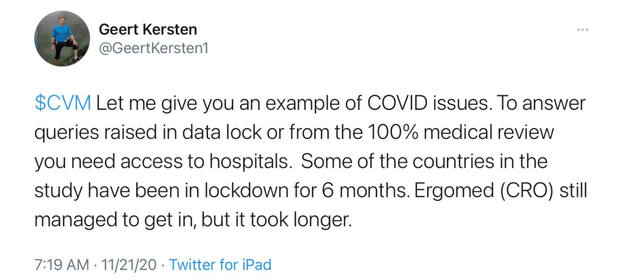
Devil is in the details. CEL-SCI doesn't say WHEN Data Lock occurred, just that its "been completed." My guess is that Data Lock occurred in Mid- November. Geert started alluding to Data Lock last month when he tweeted about Ergomed, the CRO, in past tense. See his tweet from 11/21 below:
I wrote about the prospects of Data Lock then. I was correct, again.
Why Did Data Lock Take So Long?
It took over 6 months to complete the Data Lock. The Data Lock process was impacted by COVID and the fact CEL-SCI did a Full Medical review of the data.
COVID Impact
The COVID pandemic complicated and delayed the Data Lock process. Direct access to the source data at the clinical sites was limited due to the pandemic by governments, institutions and the availability of the study personnel required to respond to any queries requiring resolution.
Geert gave additional insight into the challenges that Data Lock faced via Twitter:
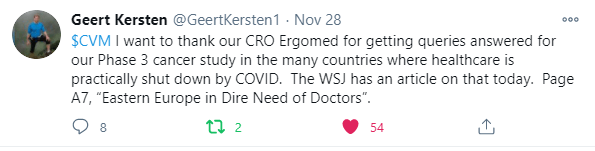
In the PR from 12/7, CEL-SCI also said "this process was particularly complicated for CEL-SCI's Phase 3 study because the study was conducted in over 20 countries on three continents, and many of these countries had, and still have, severe shutdowns due to the COVID-19 pandemic."
COVID has impacted everything in life. CEL-SCI was not immune.
Full Medical Review Impact
Furthermore, this wasn't just an ordinary Data Lock, it was a Full Medical Review. From the September Shareholder Letter:
Early this year we took one more step to ensure the completeness, accuracy, and validity of the study data. We tasked a group of physicians from Ergomed and ICON, the two CROs managing the Phase 3 study, to perform a 100% medical review of all of the study patients. That is akin to doing a 100% audit of all of the medical results."
Data Lock must be done perfectly so data can be used in support of a FDA license application. Every data point in each patient's case report form concerned with their selection, randomization, laboratory assessments, safety and efficacy evaluations of all treatments received must be reviewed, and the source data verified as complete, accurate and correct.
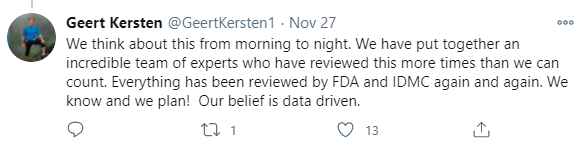
CEL-SCI erred on the side of being conservative and this required deliberate planning efforts. The data is likely to be audited by regulatory authorities prior to any license or approval being granted. The Data Lock procedures needed to be completed with extreme care before the data base could be locked and a complete analysis of the study results could be performed.
More from the recent Shareholder Letter:
The data lock process is even more complicated for our Phase 3 study because it involved three treatment arms as well as four treatment modalities, Multikine, surgery, radiation, and chemotherapy.
To be used in support of a FDA license application or a product registration anywhere in the world, every data point in each patient's case report form concerned with, among other things, their selection, randomization, laboratory assessments, safety and efficacy evaluations of all treatments received must be reviewed, and the source data verified as complete, accurate and correct. Since the data from our study will most likely be audited by regulatory authorities prior to any license or approval being granted, the data lock procedures must be completed with extreme care before the data base can finally be locked and a complete analysis of the study results can be performed. The analysis will evaluate the safety of the Multikine treatment regimen and determine if the primary, secondary and tertiary study endpoints of the Phase 3 study have been met.
The length of time it takes to lock data from a study and analyze it depends on the size and complexity of the study, the number of study sites and personnel involved, and the period of time over which the study was conducted. Our Phase 3 study was very complex and was conducted in 928 patients over the course of 9.5 years in 100 medical centers on 3 continents. The CROs have to review a lot more data for our study than in most other studies, which are not as large, not as complex or geographically dispersed, and did not run for such a long period of time.
The COVID-19 pandemic has complicated and delayed the data lock process for our Phase 3 study and added to our workload. Direct access to the source data at the clinical sites has sometimes been limited due to the pandemic by governments, institutions and the availability of the study personnel required to respond to any matters/queries requiring resolution. In some cases, and in compliance with guidance issued by the FDA and other regulatory bodies, it has been possible to perform remote data reviews and source data verification, but not in all. From what we can see the CROs are doing a good job, but everything takes longer as a consequence of the COVID-19 pandemic. The bottom line is that we have highly skilled professionals around the world working to resolve this, and they are getting it done."
But all of this is now in the past. Data is now locked and it is time to look ahead.
The Path To Full Data Announcement
Data Lock was just announced and Full Data analysis could come "very soon". CEL-SCI will continue to remain blinded to the results throughout this process.
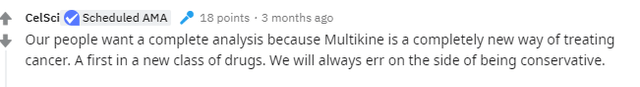
Remember, Geert has specifically stated that CEL-SCI is only presenting Full Data Analysis, not just Top Line Data. From his reddit AMA below:
 Before we look at what to expect in the coming days ahead, we first need to look back a bit.
Before we look at what to expect in the coming days ahead, we first need to look back a bit.
The Data Lock Announcement That Wasn't To Be Announced
Geert was very deliberate in his messaging about not announcing Data Lock. For example, in his AMA session on reddit, he said CEL-SCI would not announce Data Lock and they "will only announce the final data." We now know that changed.
I was vocal in the past few months that I didn't think data was locked. In addition, I expected Geert to announce Data Lock, despite many saying Geert would not announce it. I was proven correct, yet again.
I always believed that Geert was going to announce Data Lock for a few reasons. I'll never forgot the statement Geert made in the summer of 2019. Geert specifically stated CEL-SCI would not announce when the 298 event occurred. Yet, in May 2020, CEL-SCI announced that 298 had occurred.
Thus, we now have a pattern. Two significant milestones (298th event and Data Lock) were announced despite Geert saying he would not announce them. This is an important take away for the pending Full Data announcement.
Will Shorts Get Caught With Their Shorts Down?
Geert despises the Shorts. He will never, ever give them any information that would be to their advantage. He loves keeping the Shorts on their toes.
Geert's has a history of surprising Shareholders, and Shorts, by announcing information that he said he wasn't going to announce. Therefore, one would have to think twice about his comment that only Full Data Analysis, not Top Line, will be announced.
If Top Line Data is seen to be strong, say with a 15% efficacy, I wouldn't be surprised to see Geert shock the Shorts, and the World, and release positive Top Line Data prior to Full Data analysis. It would be epic.
Thus, it is my belief that some type of data announcement will come much sooner than people and the Shorts think. Since Data Locked likely occurred in Mid-November, I would not be surprised to see some type of data announcement by early January 2021!
With 10.3M shares short as of 11/13/20, this represents 28% of the float. This would be pure rocket fuel. Positive data will propel this stock the likes no one has ever seen.
The Running of The CVM Bulls
Now that Data Lock has been announced and expectations of a data announcement being "very soon", I expect a massive bull run to occur.
Let's look at what happened to another stock that recently announced Data lock: Northwest Bio (OTCQB:NWBO).
On October 5, NWBO announced Data Lock. The stock had a market cap of around $550M prior to that announcement. That amount is very similar to CEL-SCI's current market cap.
In the subsequent trading days after announcing Data Lock, NWBO's market cap ballooned to nearly a $1.9B (yes Billion) before hitting reality and settling in around $1.1B.
Thus, it's not out of the question to see CEL-SCI's market cap move up substantially over the next few weeks. A market cap of $1.1B for CEL-SCI equates to a share price of around $28. Thus, there is significant upside in the lead up to the Full Data analysis announcement.
Don't forget this excerpt from the recent Shareholder Letter. 
Geert never, ever shows his cards. Expect data sooner than you think.
BLA in Process?
It appears that CEL-SCI is no longer in a holding pattern. Clues are pointing towards a pending BLA filing. There are many steps required for a FDA Pre-Approval Inspection.
The details for a BLA filing can be found here.
Let's look at two recent clues that point towards things heating up at CEL-SCI.
New Job Postings
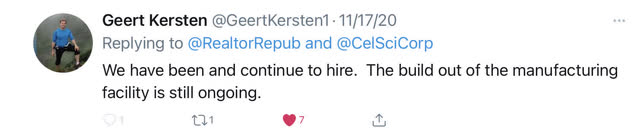
There are other clues that speak to Multikine's pending success. CEL-SCI has been hiring. Geert confirmed recently on Twitter that they continue to hire.
CEL-SCI is actively recruiting for two additional roles. On December 5, CEL-SCI's Career Page was updated with two new roles:
The two roles are:
- Quality Assurance Auditor
- Quality Assurance Document Control Assistant
Key verbiage from the postings:
Supporting the manufacture and testing of a biopharmaceutical drug."
There is no chance CEL-SCI management is incurring additional payroll expenses just to spook the Shorts. None.
In fact, there were two other job postings that are no longer on the CEL-SCI website. We can surmise that these roles have been filled:
- Manager of Bioassay
- Facility Manager
All of these positions are to get the manufacturing facility FDA compliant for the pre-approval inspection ("PAI"). There is really no other reason these positions would be announced right now. Google "FDA PAI" for yourself and I believe you will come away with the same conclusions.
Cold Fill Manufacturing Facility Upgrades
One of the many things needed for a BLA is ensuring that the manufacturing process is up to snuff.
The Cold Fill manufacturing facility is currently having modifications done to it. Per the recent 10-Q, they spent "$1.9M to prepare the manufacturing facility for the potential commercial manufacture of Multikine." However, they are not done upgrading the manufacturing facility. Geert recently tweeted this that the "build out of the manufacturing facility is still ongoing."
CEL-SCI built this facility well over 10 years ago. Some components were outdated. It requires modifications to get up to current FDA specifications.
Why spend the time, effort and money now?
It's not hard to piece all this together. The timing and roles are in sync with the theory that a BLA is being prepared.
We Believe The Trial Will Be Positive
As noted earlier, this study took nearly 10 years to complete. Over 2 years longer than anticipated.
Some readers might say that the reason for this 2 year "delay" is because Overall Survival ("OS") for head and neck cancer has improved over the years. This is categorically wrong.
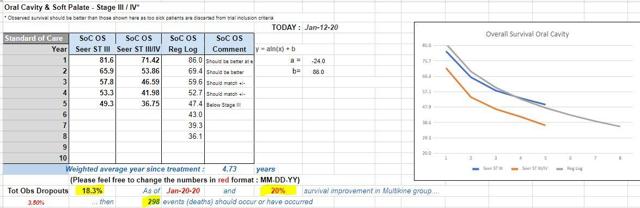
Basic internet searches will not provide the exclusion/inclusion criteria that are needed to determine the estimated OS. One must dig deep into the SEER database. CEL-SCI hired an External Statistical Group to do just that - determine the exact OS using the exact study population.
The biggest takeaways here are the "terrible" OS statistics for this study population: 3-year OS - 47% and 5-year OS - 37%. The trial assumed a survival rate of 55% at Year 3. The actual OS is 8% lower than what was estimated.
This is consistent with the American Cancer Society's declaration in January 2020:
Death rates rose over the past decade for... sites within the oral cavity and pharynx."
CEL-SCI's independent analysis is consistent with the American Cancer Society's assessment: survival for the study population has decreased and can't be the reason for the "delay".
Our own survival analysis is striking and we can't avoid the obvious. The statistics are showing an efficacy well over 10% in every one of our scenarios.
Yervoy All Over Again
Bristol-Myers Squibb (BMY) faced a similar "delay" in its Phase III trial for the blockbuster cancer immunotherapy drug Yervoy. Yervoy is an entirely different immunotherapy and indication than Multikine, but there are striking similarities. Both are Event-Driven studies, for example.
BMY became keenly aware of the challenges associated with Event-Driven studies. In February 2016, Tai-Tsang Chen, PhD Executive Director of Global Biometrics Sciences at BMY, gave a keynote presentation about Event-Driven studies.

In the presentation (Slide 10 shown above), Dr. Chen made the following observations:
- BMY anticipated that all the events were to have occurred within three years.
- Three years into its study, only 85% of the events had occurred.
- However, what is most telling is that it took two additional years for the remaining 15% events.
- Events began to trickle in and showed a very different survival curve than "standard" cancer treatments.
This is what we believe happened with Multikine: a decreasing event rate due to the "delayed clinical effect".
Risks And Conclusion
Biotechnology is risky, and so is investing in it. A majority of all biotech trials fail.
As the primary endpoint is the open door to approval by the FDA, we believe there is a very high probability of success for this drug and that CEL-SCI is currently deeply undervalued, given the potential of the drug and the probability of success. A plethora of items are pointing to success:
- Both the American Cancer Society and CEL-SCI confirm our due diligence: OS has decreased for the trial's patient population.
- Based on our statistical analysis, we expect to see an efficacy well over 10%.
- The "delayed clinical effect" has prolonged the length of the study.
- IDMC has reviewed the data and could have determined that the trial was futile, but they said to continue. They see something here.
- Dropouts are not an issue and appear to be contained.
- Other clues, like the recent new hires, insider stock purchases, lobbying efforts, upgrades to the manufacturing facility, and the large amount of in-the-money warrants, are pointing towards success.
- CEL-SCI had over $20M in cash at 6/30/20 with a burn rate of around $1.5M per month. That is well over a full year's worth of cash!
While we strongly believe that this trial should be a tremendous success, there are many aspects of a clinical trial that could be the cause of failure.
Failure could be due to many factors, such as, but not limited to:
- SOC showing an extraordinary capacity to heal patients.
- High level of dropouts during the study (well above 20%).
- Violations of the protocol not detected or reported properly by the investigator.
- Considerations due to the fact that the trial happened in many countries outside of the USA.
Speculation on this stock may be prudent from the information presented, as it seems likely this treatment will succeed. Those a little less confident may choose to go along for the ride up, if it continues, and sell as anticipation builds closer to a release of information. Those with the opposite belief may cite the company's past and obstacles to approval.
All those risks do remain, and a wise investor will consider them before making an investment decision. If the trial fails, we would expect the share price to be less than $1.
However, we believe this will succeed. We are again suggesting a fair valuation between $80 and $200 per share.
More details can be found on KillCVMShorts.com. Many thanks to my supportive friend Prestige.